From the desk of Jorge D. Faccinetti – chief editor and co-founder – I am thrilled to announce that Pituitary World News co-founder Dr. Lewis Blevins has been working on a new book on Cushing’s

News from pharmaceutical companies and other related healthcare organizations

From the desk of Jorge D. Faccinetti – chief editor and co-founder – I am thrilled to announce that Pituitary World News co-founder Dr. Lewis Blevins has been working on a new book on Cushing’s

From the desk of J D Faccinetti – co-founder – The company recently kicked off a phase 3 trial for their new nonpeptide oral drug for acromegaly and announced the dosing of their first acromegaly
Camurus AB, a pharmaceutical company based in Lund, Sweeden, is currently recruiting volunteers for two clinical trials on the long-term safety and efficacy of an octreotide subcutaneous depot to treat acromegaly. This new treatment offers

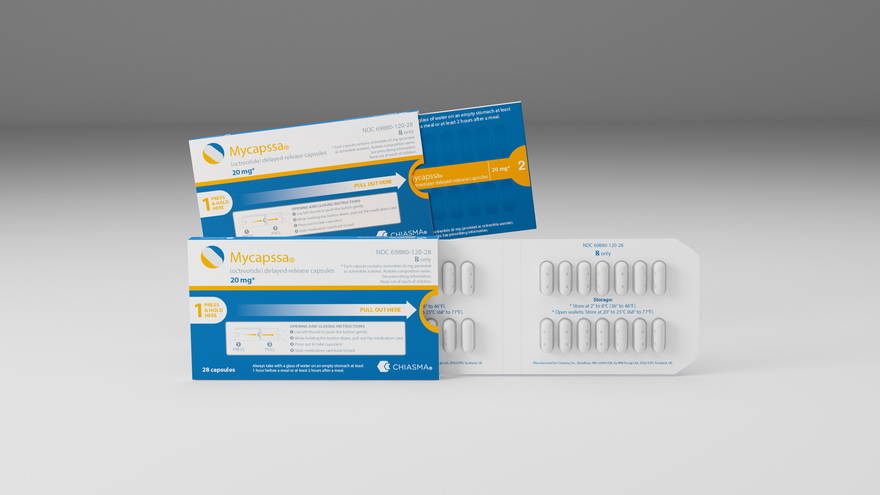
With the recent approval and commercial launch of MYCAPSSA®, Chiasma delivers the first and only oral somatostatin analog to treat acromegaly. The research showed 58% of patients maintained normal IGF-1 levels with the new oral

From a Crinetics Pharmaceutical press release: Crinetics Pharmaceuticals, Inc. (Nasdaq: CRNX), a clinical-stage pharmaceutical company focused on the discovery, development, and commercialization of novel therapeutics for rare endocrine diseases and endocrine-related tumors, announced that the

In a recent press release, Recordati Rare Diseases announced the results from a Phase III study of Isturisa® (Osilodrostat) published in Lancet Diabetes and Endocrinology. “The exciting data, published today, underscore the efficacy and safety

In a recent press release, Recordati Rare Diseases Inc., a biopharmaceutical company committed to developing therapies for rare disease communities of the United States, announced the full availability of Isturisa® for the treatment of adult

From the editors’ desk – A recent Endocrine Web article by Kimberly B. Bjugstad Ph.D. highlights a new oral octreotide drug for Acromegaly. The drug currently is on an FDA Phase III trial, referred to as OPTIMAL,

In this podcast, Dr. Lewis Blevins discusses a new drug for Cushing’s disease recently approved in the US.

In a press release today, San Diego, California based Crinetics Pharmaceuticals reports positive results from their Phase Two trial of PALTUSOTINE (the former CRN00808). This is a new oral medication to treat acromegaly and neuroendocrine

Recordati Rare Diseases, Inc., based in Lebanon, NJ, has recently acquired the rights to SIGNIFOR® LAR and the FDA approval for ISTURISA®(osilodrostat). Both products treat pituitary diseases. SIGNIFOR LAR, a “2nd generation” somatostatin analog given

If you are currently taking, or are about to start taking Lanreotide (Somatuline Depot) injections, this website from Ipsen offers a wealth of information about patient access to therapies. IPSEN CARES serves as a central